|
7/22/2023 0 Comments Radius periodic table
Ionic radius trends in the periodic table Whenever we go from left to right in transition elements series, then there happened the decrease in atomic radius for the first four members, but after that atomic radius shows systematic behavior. Atomic radius trends in transition elementsĭ and f block elements are transition elements. Shielding effect increases down the group. Number of shells increases down the group due to which the distance between outermost shell and nucleus is also increased. Why atomic radius decreases in a period?ĭue to increasing nuclear charge, the outermost orbital comes closer to the nucleus and atomic radii are decreased.Ītomic radii increase in a group from upper to downward direction. Atomic radius trends in the periodĪtomic radii decrease in a period from left to the right in the periodic table. Half of this distance is considered to be the radius of the atom.ġ Angstrom = 102 pm Atomic radius trends in the periodic tableĪtomic radii vary systematically in the periodic table. The techniques have been deployed which can measure the distance between the center of two adjacent atoms of any element. Hence, the atomic radius will also be changed. For example, if the neighboring atom is electronegative in nature then it will pull the electron more towards itself. The distribution of electrons is affected by the neighboring atoms. The reason is that the probability for finding the electron never becomes zero even if the electron is at a large distance from the nuclear. There are no sharp boundaries for the atomic orbital. The precise measurement of the size of the atom is difficult because of the following reason. Watch The recommended video to better understand atomic radius trends in periodic table The average distance between the nucleus of the atom and its outermost shell provided that the atom is assumed to be spherical is called the atomic radius. Ionization energy or ionization potential.We will discuss the following periodic properties in the modern periodic table. 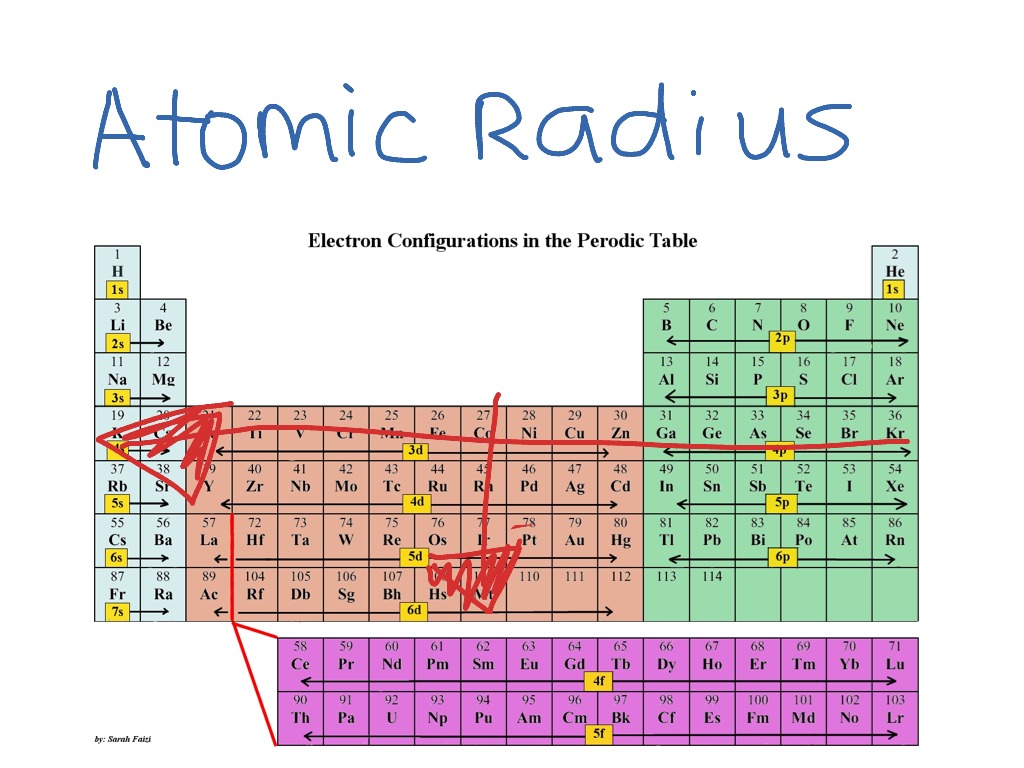
Elements are placed in groups and periods on the basis of similar properties and systematic variation of properties. In the Modern Periodic Table, the elements are arranged in the ascending order of their atomic number. It is of worth importance to know about The variation of different physical and chemical properties in the modern periodic table including groups and periods is known as the periodicity of properties. Periodic trends in the modern periodic table are the basics of chemistry. Trend of melting and boiling points in group.Variation of melting and boiling point in a group.Graphical explanation melting and boiling point in periods.Trend of melting and boiling point in periods.Periodic Trends of melting and boiling point.Non-metallic character trend in periodic table.Metallic character trend in the periodic table.Metallic character trend in periodic table.Factors affecting the electron affinity.Electron affinity trend in periodic table.Graphical explanation of ionization energy.Variation of ionization energy values in the periodic table.Factors affecting the ionization energy values.Ionization energy trend in the periodic table.Graphical representation of atomic radii.Comparative study of ionic radii vs atomic radii.Ionic radius trends in the periodic table.Atomic radius trends in transition elements.Why atomic radius decreases in a period?.Atomic radius trends in the periodic table.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
